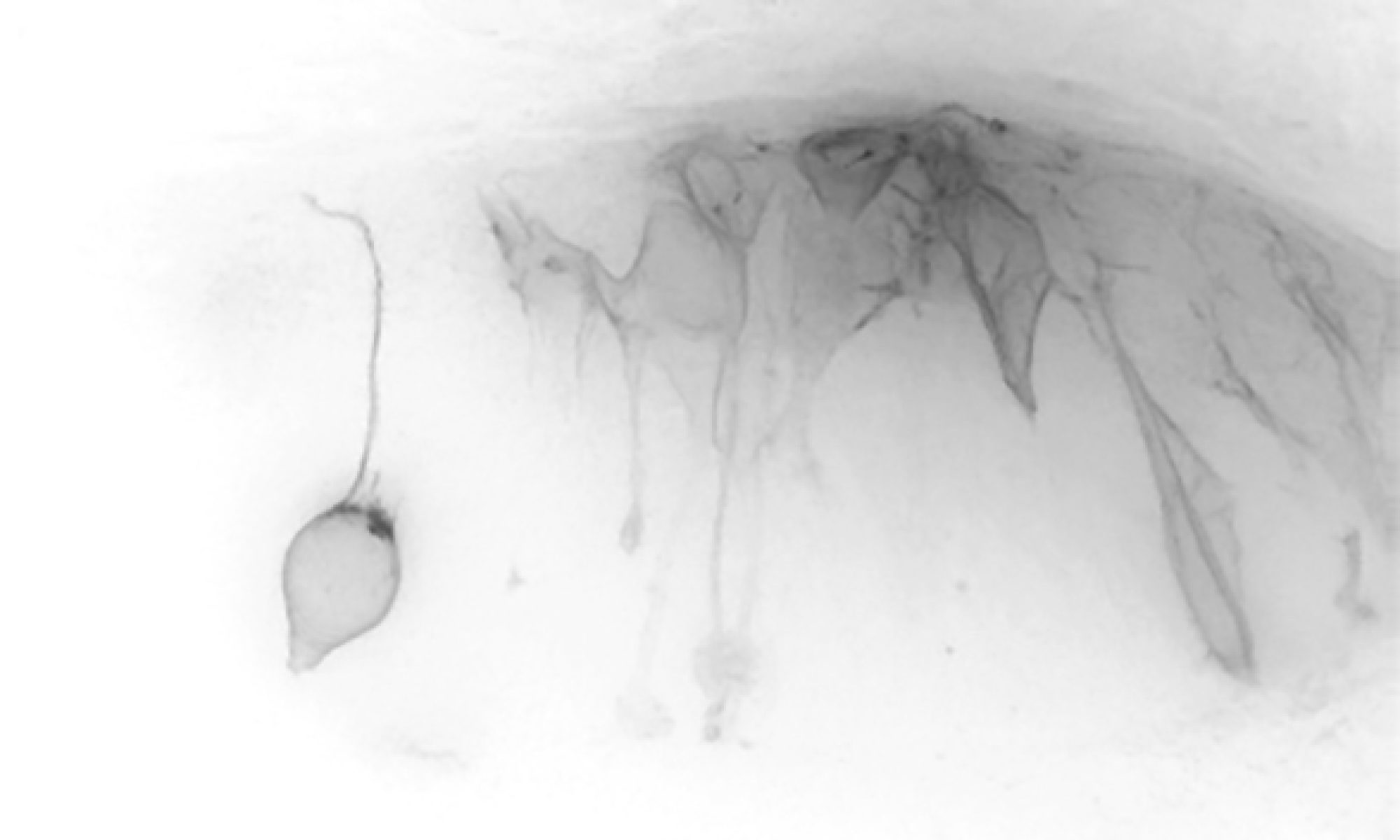
Researchers in Kate Storey’s laboratory investigate how the nervous system forms during embryonic development.
We all come from a single cell, the fertilised egg. As this cell divides to generate the embryo individual cells begin to specialise in a process known as cellular differentiation.
We wish to understand how neural cells arise in the embryo and how their differentiation is controlled.
The spinal cord is generated progressively as the embryonic body is laid down and this helps us to identify and investigate the distinct steps that underlie its formation. These include when and how neural precursor cells stop dividing and become nerve cells, and how these cells then change shape and ultimately make connections to form functional neural circuitry.
Our team uses a wide range of techniques, including gain and loss of gene function approaches and genome-wide analyses, as well as live imaging assays, which allow us to monitor changes in cell behaviour during differentiation.
Our overall aim is to discover cellular and molecular mechanisms that control neural differentiation. Elucidating these mechanisms helps us to understand better how the developing nervous system is impacted by cellular stress and may inform strategies for therapeutic treatment of neural injury and disease.